Carbohydrates
Introduction:
Carbohydrates are essential biomolecules that provide energy, support metabolic activities, and form important structural components of cells. They are a major part of the human diet and are commonly found in foods such as rice, bread, fruits, milk, and vegetables. A clear understanding of carbohydrates is important for pharmacy students studying biochemistry, clinical pathology, and nutrition.
These notes explain carbohydrates in a simple, student-friendly manner suitable for D.Pharmacy and B.Pharmacy students.
Definition of Carbohydrates:
Carbohydrates are organic compounds composed of carbon, hydrogen, and oxygen. Their general chemical formula is Cn(H2O)n. They are commonly referred to as sugars or saccharides.
In simple words, carbohydrates are the primary fuel molecules of the body.
Classification of Carbohydrates:
Carbohydrates are classified based on the number of sugar units present in their structure.
1. Monosaccharides (Single Sugar Units):
- Glucose: Found in blood and fruits
- Fructose: Known as fruit sugar
- Galactose: Component of milk sugar
2. Disaccharides (Two Sugar Units):
- Maltose: Glucose + Glucose
- Lactose: Glucose + Galactose
- Sucrose: Glucose + Fructose
3. Polysaccharides (Many Sugar Units):
- Starch: Storage carbohydrate in plants
- Glycogen: Storage carbohydrate in humans and animals
Chemical Properties of Carbohydrates:
- Reducing Property: Reducing sugars like glucose and lactose reduce Benedict’s and Fehling’s solutions.
- Oxidation: Carbohydrates form acids on oxidation.
- Fermentation: Glucose is converted into alcohol and carbon dioxide by yeast.
- Dehydration: Concentrated acids convert carbohydrates into carbon or furfural derivatives.
- Formation of Esters and Ethers: Hydroxyl groups of sugars react with acids and alkyl groups.
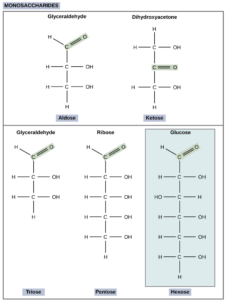
Monosaccharides:
Monosaccharides are the simplest form of carbohydrates and cannot be hydrolyzed into smaller sugar units.
Structure of Glucose:
Glucose (C6H12O6) is an aldohexose. It exists in both open-chain and ring forms. The ring structure is a six-membered ring known as pyranose. Glucose is the main sugar present in blood.
Structure of Fructose:
Fructose is a ketohexose that forms a five-membered ring called furanose. It is the sweetest naturally occurring sugar and is found in fruits and honey.
Structure of Galactose:
Galactose is an aldohexose similar to glucose but differs in the orientation of one hydroxyl group. It is a component of lactose (milk sugar).
Disaccharides:
Disaccharides are formed when two monosaccharides are linked together by a glycosidic bond.
Maltose:
- Structure: Glucose + Glucose
- Formed during digestion of starch
- Reducing sugar
Lactose:
- Structure: Glucose + Galactose
- Present in milk
- Reducing sugar
Sucrose:
- Structure: Glucose + Fructose
- Common table sugar
- Non-reducing sugar
Polysaccharides:
Starch:
Starch is the major storage carbohydrate in plants. It consists of:
- Amylose: Linear chain
- Amylopectin: Branched chain
During digestion, starch is broken down into glucose.
Glycogen:
Glycogen is the storage carbohydrate in humans and animals. It is stored mainly in the liver and muscles. It has a highly branched structure, allowing rapid release of glucose when required.
Qualitative Tests for Carbohydrates:
- Molisch’s Test: General test; violet ring indicates presence of carbohydrates.
- Benedict’s Test: Brick-red precipitate indicates reducing sugars.
- Fehling’s Test: Formation of red cuprous oxide precipitate.
- Barfoed’s Test: Monosaccharides react faster than disaccharides.
- Iodine Test: Starch gives blue-black color; glycogen gives reddish-brown color.
Biological Roles of Carbohydrates:
- Main Source of Energy: Provides 4 kcal per gram.
- Energy Storage: Stored as glycogen in liver and muscles.
- Structural Role: Ribose and deoxyribose form RNA and DNA.
- Protein Sparing Action: Prevents protein breakdown for energy.
- Maintains Blood Glucose: Essential for brain and RBC function.
- Fat Metabolism: Necessary for proper utilization of fats.
Summary:
Carbohydrates are vital biomolecules that serve as the primary energy source and play key roles in metabolism and cellular structure. For pharmacy students, understanding their classification, properties, tests, and biological roles is essential for studying biochemistry, clinical diagnosis, and drug therapy.
Frequently Asked Questions (FAQs)
1. What are carbohydrates in simple terms?
Carbohydrates are organic compounds that provide energy to the body and are commonly known as sugars or starches.
2. Why are carbohydrates important for the human body?
They are the main source of energy and help maintain blood glucose levels, especially for the brain.
3. What is the difference between monosaccharides and polysaccharides?
Monosaccharides contain a single sugar unit, while polysaccharides consist of many sugar units linked together.
4. Which carbohydrates are reducing sugars?
Glucose, lactose, maltose, and other sugars with free aldehyde or ketone groups are reducing sugars.
5. Why is glycogen important?
Glycogen acts as a stored form of glucose in the liver and muscles, providing energy during fasting or physical activity.
Dr. Saint Paul is a pharmacy educator, Pharm.D graduate, and academic content creator from Jawaharlal Nehru Technological University Kakinada (JNTUK), where he completed his Doctor of Pharmacy (Pharm.D) degree between 2015 and 2021.
He has more than 7 years of experience creating pharmacy educational content, writing study materials, and reviewing academic articles for pharmacy students. He has also contributed guest articles to pharmacy education platforms, including PharmD Guru.
At D.PharmGuru, his work focuses on simplifying complex Diploma in Pharmacy (D.Pharmacy) subjects into easy-to-understand notes, practical explanations, and exam-oriented educational resources for students across India.
His areas of focus include Human Anatomy and Physiology, Pharmaceutics, Pharmacology, Pharmaceutical Chemistry, Hospital and Clinical Pharmacy, and other core D.Pharmacy subjects.



